
The suffix changes to ‘–ite’ with one less oxygen, or it is given the prefix ‘per–’ with one oxygen atom more. The most common oxyanion of an element is named by combining the element’s base-name with the suffix ‘–ate’. Polyatomic anions containing oxygen, or oxyanions, are named based on the number of oxygen atoms.įor example, multiple polyatomic ionic compounds are formed between sodium and oxyanions of bromine. Polyatomic ionic compounds follow the same nomenclature rules, but apply the polyatomic anion name instead of the suffix ‘-ide’. Thus, the names are sodium iodide, iron(II) bromide, and iron(III) bromide. Iron forms two types of cations, which are displayed using parenthesized Roman-numerals. Since sodium forms only a single type of cation, the parenthesized Roman-numeral is excluded. The anion base-name ending with the suffix ‘–ide’ follows last.Ĭonsider the following three molecules. The chemical formula helps determine the name of the compound.īinary ionic compounds name the metal cation first, succeeded by a parenthesized Roman-numeral indicating the metal charge if the metal can exist in different cationic states. The cationic subscript is numerically matched to the anionic charge and vice versa, generating a charge-neutral chemical formula.Īll ionic compounds are named by following a systematic naming convention called the chemical nomenclature. Next, the overall charge within the formula is balanced by employing a crisscross method. Conversely, fluorine from group 17 forms anions with an ionic-charge of 1−. Calcium from group 2 forms cations with an ionic-charge of 2+. Ionic charges can be determined from the group number of the element in the periodic table. 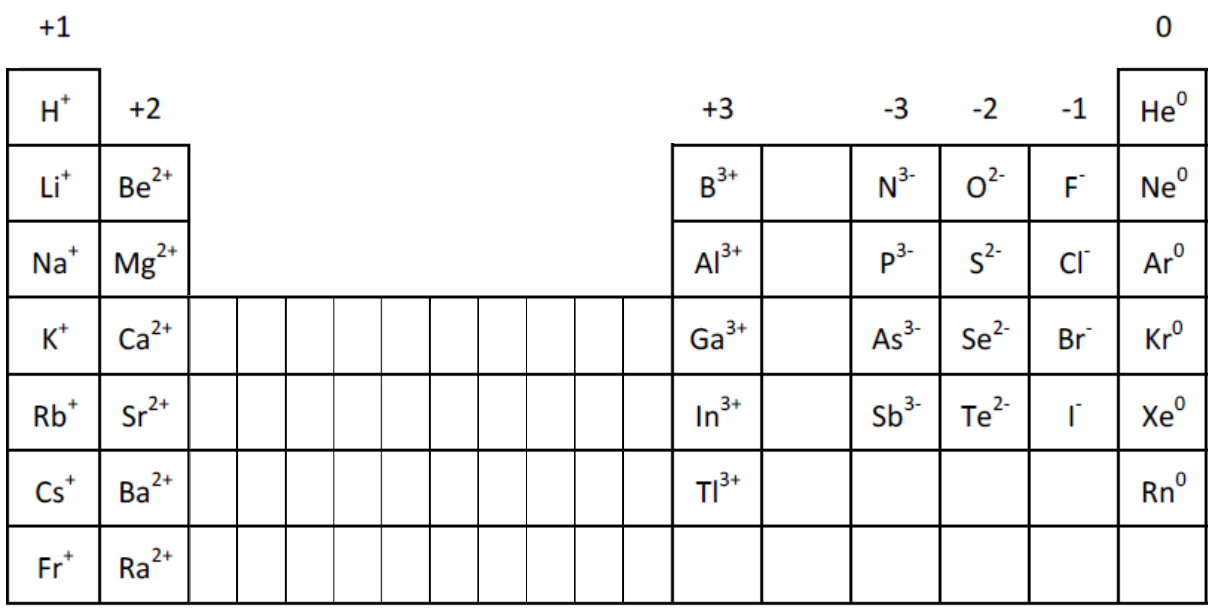
Start with the cations followed by anions. First, specify the ions of each element and their respective ionic charges. These compounds are represented by chemical formulas.Ĭonsider calcium fluoride. 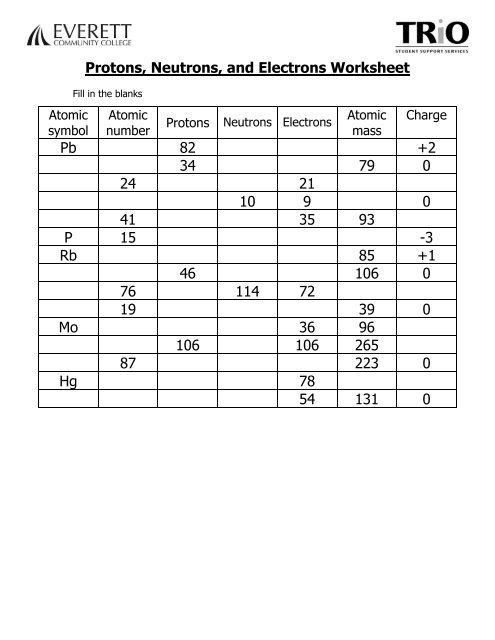
Ionic compounds form ionic bonds between positively charged metal ions, or cations, and negatively charged nonmetal ions, or anions.
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
